Oculus Spirit [98609]
TigerPulse: 100%
Posts: 65375
Joined: 7/13/02
|
When a medical expert says "there is no evidence" that
Jan 24, 2023, 12:23 PM
|
|
covid damages your immune system, just know "no evidence" is a giant red flag. You seek no evidence, so you get no evidence, so you can say there is no evidence, so someone thinks it isn't so, when it is. That's how "no evidence" works. Keep that in mind. This goes beyond covid. Politics, whatever, when you hear someone say "there is no evidence", that is a hedge for them to make you think/assume a negative, when in reality it is a ploy the speaker uses to justify misinformation by feigning ignorance. When confronted with evidence, the speaker can then say "How could we have known", based on no evidence, when there was evidence they chose to ignore to say there is no evidence. It's an obtuse, convoluted rhetorical technique, but you have to recognize it when you see it.
Oh, below is some "no evidence"....
https://pubmed.ncbi.nlm.nih.gov/32425950/
https://www.researchgate.net/publication/342997179_AIDS_and_COVID-19_are_two_diseases_separated_by_a_common_lymphocytopenia
https://www.medrxiv.org/content/10.1101/2020.09.25.20200329v1.full.pdf
https://www.pnas.org/doi/full/10.1073/pnas.2010722117
https://www.nature.com/articles/s41467-020-19706-9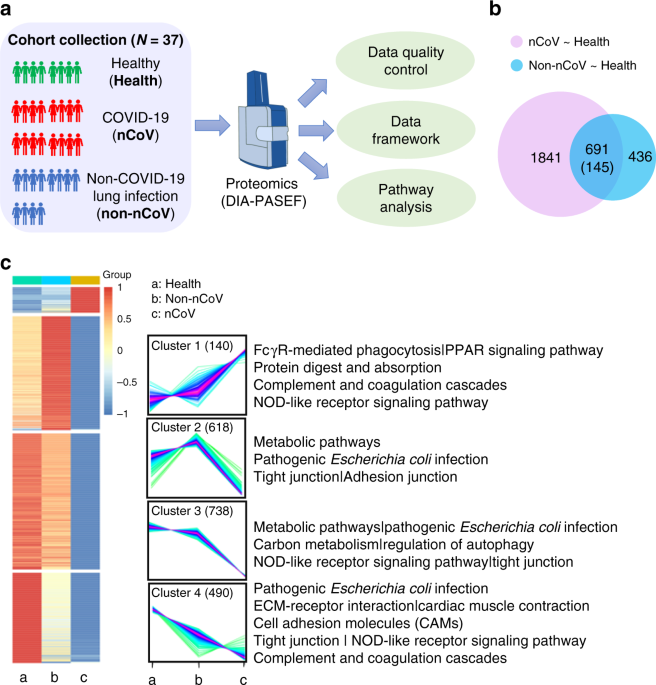 Immune suppression in the early stage of COVID-19 disease - Nature Communications The outbreak of COVID-19 has become a worldwide pandemic. The pathogenesis of this infectious disease and how it differs from other drivers of pneumonia is unclear. Here we analyze urine samples from COVID-19 infection cases, healthy donors and non-COVID-19 pneumonia cases using quantitative proteomics. The molecular changes suggest that immunosuppression and tight junction impairment occur in the early stage of COVID-19 infection. Further subgrouping of COVID-19 patients into moderate and severe types shows that an activated immune response emerges in severely affected patients. We propose a two-stage mechanism of pathogenesis for this unusual viral infection. Our data advance our understanding of the clinical features of COVID-19 infections and provide a resource for future mechanistic and therapeutics studies. How COVID-19 pathology differs from other drivers of pneumonia is unclear. Here the authors analyze urine from patients with COVID-19 and identify an immunosuppressive protein expression pattern that is distinct from the pattern in healthy individuals or patients with non-COVID-19 pneumonia.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7779612/
https://www.jci.org/articles/view/146614
https://www.nature.com/articles/s41392-021-00749-3 SARS-CoV-2 infection causes immunodeficiency in recovered patients by downregulating CD19 expression in B cells via enhancing B-cell metabolism - Signal Transduction and Targeted Therapy The SARS-CoV-2 infection causes severe immune disruption. However, it is unclear if disrupted immune regulation still exists and pertains in recovered COVID-19 patients. In our study, we have characterized the immune phenotype of B cells from 15 recovered COVID-19 patients, and found that healthy controls and recovered patients had similar B-cell populations before and after BCR stimulation, but the frequencies of PBC in patients were significantly increased when compared to healthy controls before stimulation. However, the percentage of unswitched memory B cells was decreased in recovered patients but not changed in healthy controls upon BCR stimulation. Interestingly, we found that CD19 expression was significantly reduced in almost all the B-cell subsets in recovered patients. Moreover, the BCR signaling and early B-cell response were disrupted upon BCR stimulation. Mechanistically, we found that the reduced CD19 expression was caused by the dysregulation of cell metabolism. In conclusion, we found that SARS-CoV-2 infection causes immunodeficiency in recovered patients by downregulating CD19 expression in B cells via enhancing B-cell metabolism, which may provide a new intervention target to cure COVID-19.
https://www.mdpi.com/2076-0817/10/10/1267/htm Uncertainty around the Long-Term Implications of COVID-19 Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has infected more than 231 million people globally, with more than 4.7 million deaths recorded by the World Health Organization as of 26 September 2021. In response to the pandemic, some countries (New Zealand, Vietnam, Taiwan, South Korea and others) have pursued suppression strategies, so-called Zero COVID policies, to drive and maintain infection rates as close to zero as possible and respond aggressively to new cases. In comparison, European countries and North America have adopted mitigation strategies (of varying intensity and effectiveness) that aim primarily to prevent health systems from being overwhelmed. With recent advances in our understanding of SARS-CoV-2 and its biology, and the increasing recognition there is more to COVID-19 beyond the acute infection, we offer a perspective on some of the long-term risks of mutational escape, viral persistence, reinfection, immune dysregulation and neurological and multi-system complications (Long COVID).
https://insight.jci.org/articles/view/146701
https://www.nature.com/articles/s41590-021-01113-x?s=09
https://www.frontiersin.org/articles/10.3389/fimmu.2022.853606/full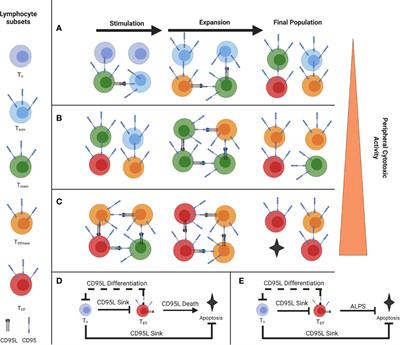 Understanding the Effects of Age and T-Cell Differentiation on COVID-19 Severity: Implicating a Fas/FasL-mediated Feed-Forward Controller of T-Cell Differentiation Abstract Fas expression is quickly upregulated on CD8+ T cells following stimulation, while FasL expression is limited to Tcm and Tem.1 A phenomenon of T cell effector differentiation via paracrine Fas-FasL signal has been previously described, along with evidence of T-T interactions that drive cytotoxic function.1–3 Here, we describe such T-T interaction and differentiation where the lymphocytes perceive the phenotypic density of their own populations and make unified group decisions as quorum sensing, and propose a Feed-forward model which can prototypically correct for disturbances in the system, as seen during in vitro T cell stimulation.1,4–8 This feed-forward controller exerts control via early Tn Fas expression, memory/Effector FasL expression, and apoptosis at terminal differentiation, all mediated through a single, dual-purpose protein with two signaling functions, differentiation and death, which are mutagenically uncoupled by the C194V mutation in mice and in Autoimmune Lymphoproliferative Syndrome (ALPS) patients.9 Interestingly, the feed-forward model provides us with evolutionary insight as to why Fas stimulation becomes apoptotic at terminal differentiation, and it is useful in order to reconcile the wide discrepancy between youth and adult Covid-19 severity.10,11 Introduction Feed-forward loops are means of managing homeostasis under dynamic conditions.4–6 It is a way for organisms and cells to manage inputs and responses.4 Feed-forward control differs from...
https://www.science.org/doi/10.1126/science.abm8108
https://www.biorxiv.org/content/10.1101/2022.04.07.487556v1.full.pdf
https://www.nature.com/articles/s41392-022-00919-x#citeas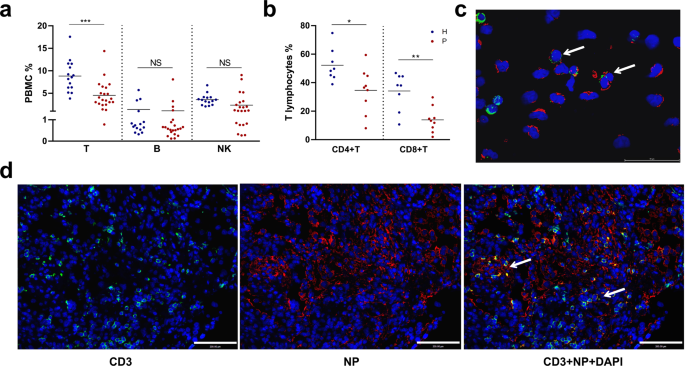 ACE2-independent infection of T lymphocytes by SARS-CoV-2 - Signal Transduction and Targeted Therapy SARS-CoV-2 induced marked lymphopenia in severe patients with COVID-19. However, whether lymphocytes are targets of viral infection is yet to be determined, although SARS-CoV-2 RNA or antigen has been identified in T cells from patients. Here, we confirmed that SARS-CoV-2 viral antigen could be detected in patient peripheral blood cells (PBCs) or postmortem lung T cells, and the infectious virus could also be detected from viral antigen-positive PBCs. We next prove that SARS-CoV-2 infects T lymphocytes, preferably activated CD4 + T cells in vitro. Upon infection, viral RNA, subgenomic RNA, viral protein or viral particle can be detected in the T cells. Furthermore, we show that the infection is spike-ACE2/TMPRSS2-independent through using ACE2 knockdown or receptor blocking experiments. Next, we demonstrate that viral antigen-positive T cells from patient undergone pronounced apoptosis. In vitro infection of T cells induced cell death that is likely in mitochondria ROS-HIF-1a-dependent pathways. Finally, we demonstrated that LFA-1, the protein exclusively expresses in multiple leukocytes, is more likely the entry molecule that mediated SARS-CoV-2 infection in T cells, compared to a list of other known receptors. Collectively, this work confirmed a SARS-CoV-2 infection of T cells, in a spike-ACE2-independent manner, which shed novel insights into the underlying mechanisms of SARS-CoV-2-induced lymphopenia in COVID-19 patients.
https://onlinelibrary.wiley.com/doi/10.1111/all.15372
https://www.biorxiv.org/content/10.1101/2022.05.20.492764v1.full.pdf
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9057012/
https://www.biorxiv.org/content/10.1101/2022.08.18.504053v1.full.pdf
https://academic.oup.com/cid/article/75/7/1232/6572226?login=false
https://www.biorxiv.org/content/10.1101/2022.10.18.512708v1.full.pdf
https://www.frontiersin.org/articles/10.3389/fimmu.2021.676932/full Longitudinal Analysis of COVID-19 Patients Shows Age-Associated T Cell Changes Independent of Ongoing Ill-Health ObjectivesThe immunological and inflammatory changes following acute COVID-19 are hugely variable. Persistent clinical symptoms following resolution of initial infection, termed long COVID, are also hugely variable, but association with immunological changes has not been described. We investigate changing immunological parameters in convalescent COVID-19 and interrogate their potential relationships with persistent symptoms.MethodsWe performed paired immunophenotyping at initial SARS-CoV-2 infection and convalescence (n=40, median 68 days) and validated findings in 71 further patients at median 101 days convalescence. Results were compared to 40 pre-pandemic controls. Fatigue and exercise tolerance were assessed as cardinal features of long COVID using the Chalder Fatigue Scale and 6-minute-walk test. The relationships between these clinical outcomes and convalescent immunological results were investigated.ResultsWe identify persistent expansion of intermediate monocytes, effector CD8+, activated CD4+ and CD8+ T cells, and reduced naïurl=https://www.frontiersin.org/articles/10.3389/fimmu.2021.676932/40, median 68 days) and validated findings in 71 further patients at median 101 days convalescence. Results were compared to 40 pre-pandemic controls. Fatigue and exercise tolerance were assessed as cardinal features of long COVID using the Chalder Fatigue Scale and 6-minute-walk test. The relationships between these clinical outcomes and convalescent immunological results were investigated.ResultsWe identify persistent expansion of intermediate monocytes, effector CD8+, activated CD4+ and CD8+ T cells, and reduced naïve CD4+ and CD8+ T cells at 68 days, with activated CD8+ T cells remaining increased at 101 days. Patients >60 years also demonstrate reduced naïve CD4+ and CD8+ T cells and expanded activated CD4+ T cells at 101 days. Ill-health, fatigue, and reduced exercise tolerance were common in this cohort. These symptoms were not associated with immune cell populations or circulating inflammatory cytokines.ConclusionWe demonstrate myeloid recovery but ...
|
|


 to award
the award.
to award
the award.












